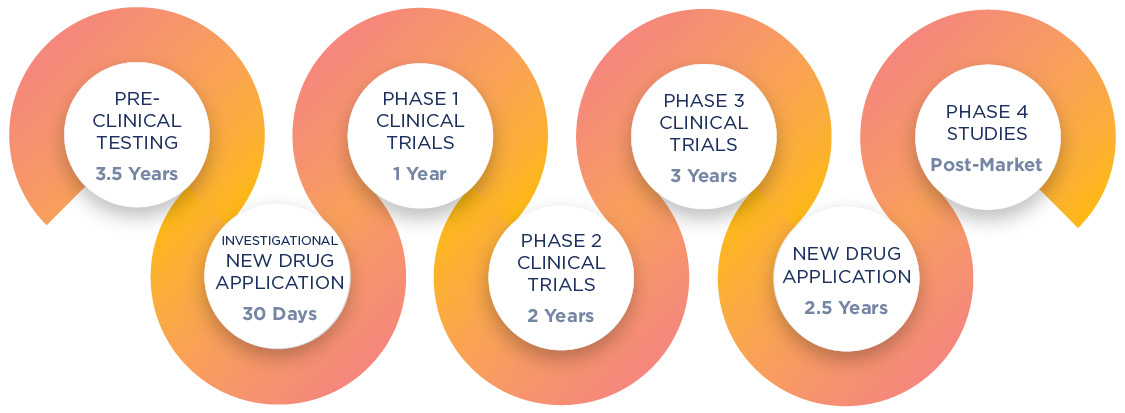
Datroway (datopotamab deruxtecan-dlnk) Lyophilized Powder for Injection
Company: AstraZeneca and Daiichi Sankyo
Date of Approval: January 17, 2025
Treatment for: Breast Cancer
Datroway (datopotamab deruxtecan-dlnk) is a TROP2-directed antibody and topoisomerase inhibitor conjugate used for the treatment of hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer.
- FDA Approves Datroway (datopotamab deruxtecan-dlnk) for Patients with Previously Treated Metastatic HR Positive, HER2 Negative Breast Cancer – January 17, 2025
- Datroway FDA Approval History
Grafapex (treosulfan) Lyophilized Powder for Injection
Company: Medexus Pharmaceuticals Inc.
Date of Approval: January 21, 2025
Treatment for: Stem Cell Transplant Conditioning
Grafapex (treosulfan) is an alkylating agent indicated in combination with fludarabine as a preparative regimen for allogeneic hematopoietic stem cell transplantation in adult and pediatric patients one year of age and older with acute myeloid leukemia (AML) or myelodysplastic syndrome (MDS).
- FDA Approves Grafapex (treosulfan) Preparative Regimen for Allogeneic Hematopoietic Stem Cell Transplantation – January 23, 2025
- Grafapex FDA Approval History
Avtozma (tocilizumab-anoh) Injection
Company: Celltrion USA
Date of Approval: January 24, 2025
Treatment for: Rheumatoid Arthritis, Giant Cell Arteritis, Polyarticular Juvenile Idiopathic Arthritis, Juvenile Rheumatoid Arthritis, COVID-19
Avtozma (tocilizumab-anoh) is an interleukin-6 (IL-6) receptor antagonist biosimilar to Actemra used for treatment of rheumatoid arthritis, giant cell arteritis, polyarticular juvenile idiopathic arthritis, systemic juvenile idiopathic arthritis, and COVID-19.
- FDA Approves Avtozma (tocilizumab-anoh), a Biosimilar to Actemra – January 30, 2025
- Avtozma FDA Approval History
Symbravo (meloxicam and rizatriptan) Tablets – formerly AXS-07
Company: Axsome Therapeutics, Inc. Date of Approval: January 30, 2025 Treatment for: Migraine
Symbravo (meloxicam and rizatriptan) is a non-steroidal anti-inflammatory drug and triptan combination indicated for the acute treatment of migraine with or without aura in adults.
- FDA Approves Symbravo (meloxicam and rizatriptan) for the Acute Treatment of Migraine with or without Aura in Adults – January 31, 2025
- Symbravo FDA Approval History
Journavx (suzetrigine) Tablets – formerly VX-548
Company: Vertex Pharmaceuticals Incorporated Date of Approval: January 30, 2025 Treatment for: Pain
Journavx (suzetrigine) is a sodium channel blocker indicated for the treatment of moderate to severe acute pain in adults.
- FDA Approves Journavx (suzetrigine), a First-in-Class Treatment for Adults With Moderate-to-Severe Acute Pain – January 30, 2025
- Journavx FDA Approval History
Onapgo (apomorphine hydrochloride) Subcutaneous Infusion Device – formerly SPN-830
Company: Supernus Pharmaceuticals, Inc. Date of Approval: February 3, 2025 Treatment for: Parkinson’s Disease
Onapgo (apomorphine hydrochloride) is a continuous subcutaneous infusion formulation of the approved dopaminergic agonist apomorphine for the treatment of motor fluctuations (OFF episodes) in adults with advanced Parkinson’s disease.
- FDA Approves Onapgo (apomorphine hydrochloride) for the Treatment of Motor Fluctuations in Adults with Advanced Parkinson’s Disease – February 4, 2025
- Onapgo FDA Approval History

Emblaveo (avibactam and aztreonam) Lyophilized Powder for Injection
Company: AbbVie Inc. Date of Approval: February 7, 2025 Treatment for: Intraabdominal Infection
Emblaveo (aztreonam and avibactam) is a monobactam antibacterial and beta-lactamase inhibitor combination used for the treatment of complicated intra-abdominal infections.
- FDA Approves Emblaveo (aztreonam and avibactam) for the Treatment of Adults With Complicated Intra-Abdominal Infections – February 7, 2025
- Emblaveo FDA Approval History
Gomekli (mirdametinib) Capsules and Tablets for Oral Suspension
Company: SpringWorks Therapeutics, Inc. Date of Approval: February 11, 2025 Treatment for: Neurofibromatosis
Gomekli (mirdametinib) is a MEK inhibitor for the treatment of patients with neurofibromatosis type 1-associated plexiform neurofibromas (NF1-PN).
- FDA Approves Gomekli (mirdametinib) for the Treatment of Adult and Pediatric Patients with NF1-PN – February 11, 2025
- Gomekli FDA Approval History

Xbryk (denosumab-dssb) Injection
Company: Samsung Bioepis Co., Ltd. Date of Approval: February 13, 2025 Treatment for: Osteolytic Bone Lesions of Multiple Myeloma, Osteolytic Bone Metastases of Solid Tumors, Giant Cell Tumor of Bone, Hypercalcemia of Malignancy
Xbryk (denosumab-dssb) is a RANK ligand (RANKL) inhibitor biosimilar to Xgeva (denosumab) for use in the prevention of skeletal-related events in patients with multiple myeloma or bone metastases from solid tumors, treatment of giant cell tumor of bone, and treatment of hypercalcemia of malignancy.
- FDA Approves Xbryk (denosumab-dssb), a Biosimilar to Xgeva – February 16, 2025
- Xbryk FDA Approval History
Ospomyv (denosumab-dssb) Injection
Company: Samsung Bioepis Co., Ltd. Date of Approval: February 13, 2025 Treatment for: Osteoporosis
Ospomyv (denosumab-dssb) is a RANK ligand (RANKL) inhibitor biosimilar to Prolia (denosumab) used in the treatment of osteoporosis.
- FDA Approves Ospomyv (denosumab-dssb), a Biosimilar to Prolia – February 16, 2025
- Ospomyv FDA Approval History