1. New Drug Application (NDA)
- Used for brand-new drugs that have not been previously approved.
- Requires clinical trial data demonstrating safety and efficacy.
- Can be classified as:
- 505(b)(1) NDA – For completely new drug entities.
- 505(b)(2) NDA – For drugs that rely on some existing data (e.g., reformulations, new dosages).
2. Abbreviated New Drug Application (ANDA)
- Used for generic drugs that are bioequivalent to already approved brand-name drugs.
- Does not require new clinical trials, just proof of bioequivalence.
- Approved under the Hatch-Waxman Act.
3. Biologics License Application (BLA)
- For biological products like vaccines, monoclonal antibodies, and gene therapies.
- Evaluated by the FDA’s Center for Biologics Evaluation and Research (CBER).
Therapeutic Biologics Applications (BLA)
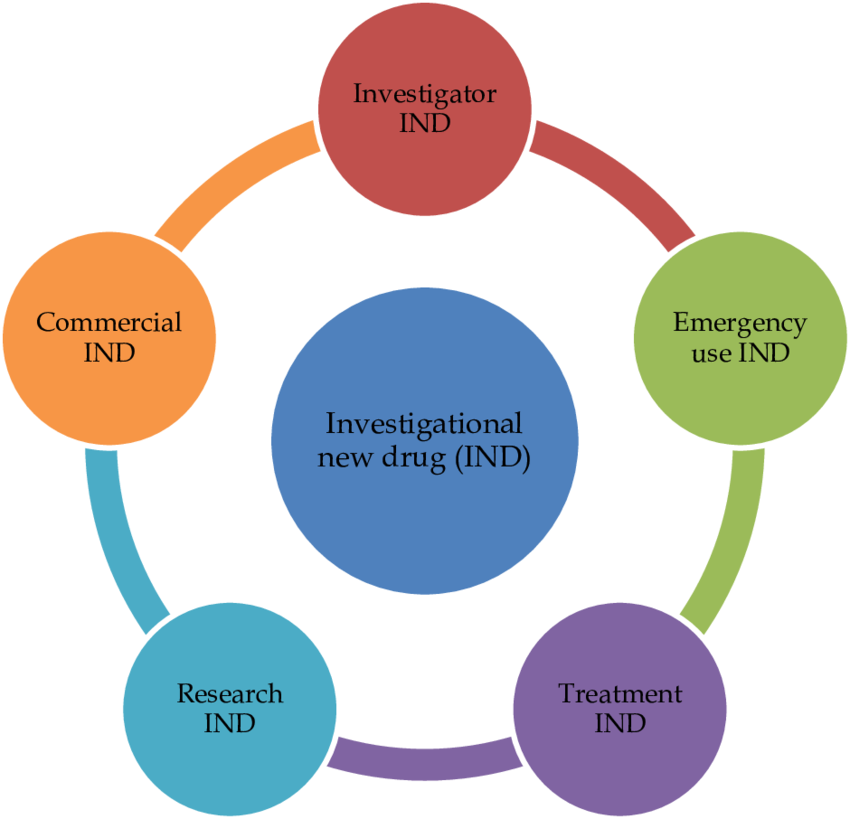
4. Investigational New Drug (IND) Application
- Required before clinical trials can begin.
- Allows testing of experimental drugs on humans.
- Types:
- Commercial IND – For companies developing new drugs.
- Research IND – For academic or non-commercial research
Investigational New Drug (IND) Application

5. Over-the-Counter (OTC) Drug Application
- Used to switch prescription drugs to OTC status.
- Requires proof of safety for self-administration without a doctor’s supervision.
6. Supplemental New Drug Application (sNDA)
- Submitted for modifications to an already approved drug.
- Changes may include new indications, new dosage forms, labeling updates, etc.
7. Emergency Use Authorization (EUA)
- Allows temporary approval of drugs or vaccines during public health emergencies (e.g., COVID-19 vaccines).
- Requires less extensive data than a full NDA or BLA.
Related Products
-
Tapentadol 100 mg
$2.00 / Per Pill
-
Fenbendazole 500mg
$3.00 / Per Pill
-
Ivermectin 12mg
$2.00 / Per Pill