Vaccine Production and Distribution: A Comprehensive Overview
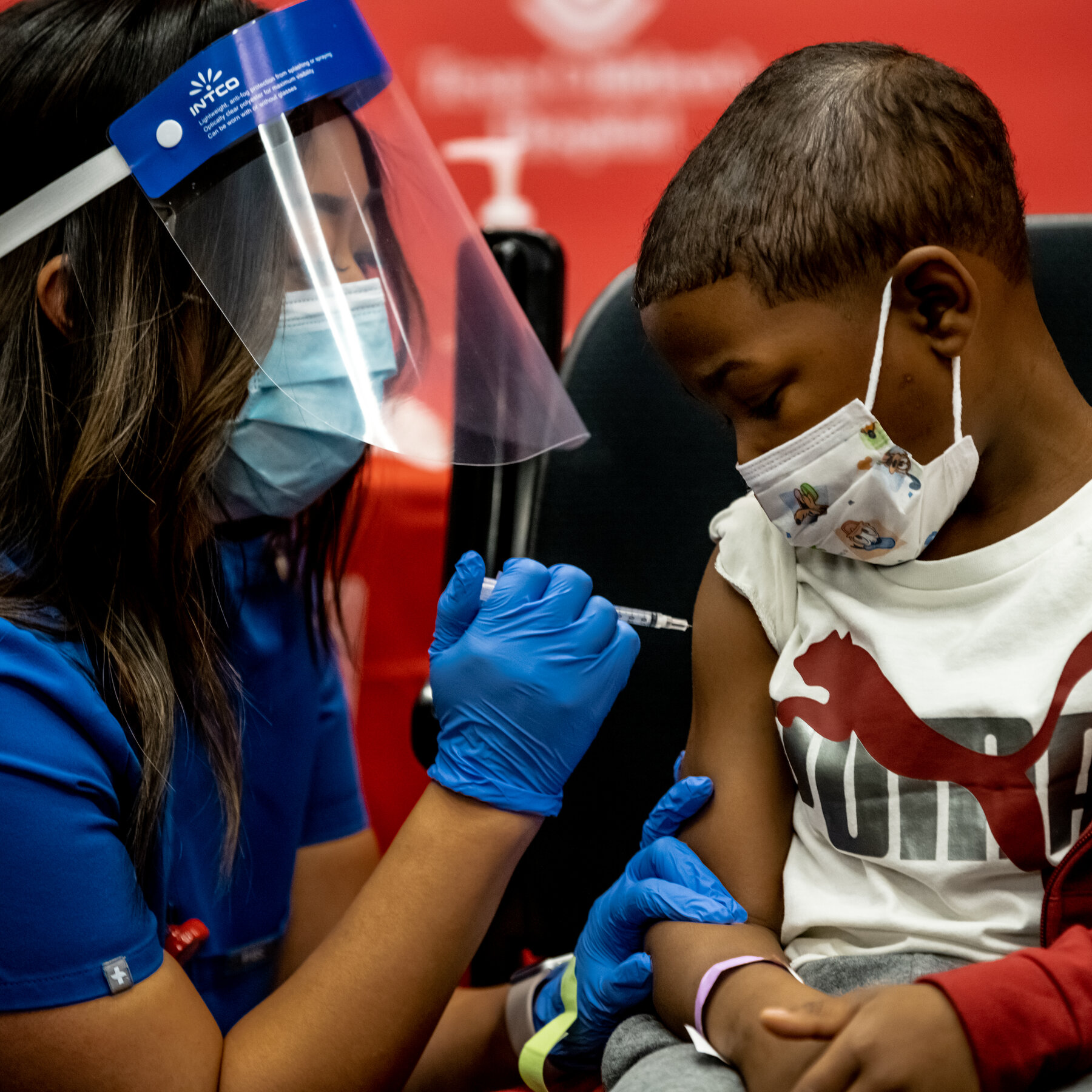
Vaccine production and distribution is a critical aspect of global public health, aiming to prevent the spread of infectious diseases and protect populations from potential outbreaks. The process is complex and multi-phased, involving scientific research, stringent testing, regulatory approvals, large-scale manufacturing, and widespread logistical coordination.
Vaccine Development and Production
The journey of a vaccine begins in the laboratory. Scientists first identify the pathogen—typically a virus or bacterium—and determine which components of it can safely trigger an immune response. These components could be whole inactivated pathogens, protein subunits, viral vectors, or messenger RNA (mRNA), as seen with recent COVID-19 vaccines.
Once a vaccine candidate is developed, it undergoes preclinical testing in cell cultures and animal models. If the results are promising, clinical trials in humans begin. These trials occur in three phases:
Phase I tests safety in a small group of healthy volunteers.
Phase II expands the trial to a larger group to further assess safety and immune response.
Phase III involves thousands of participants to evaluate efficacy and monitor for adverse effects.
Only after passing all phases does a vaccine receive approval from regulatory bodies like the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), or the World Health Organization (WHO).
Once approved, vaccines move into full-scale production. Manufacturing is conducted in specialized facilities with high biosafety standards. Quality control is critical—each batch is rigorously tested to ensure it meets safety, potency, and purity standards. Manufacturing can take weeks to months, depending on the type of vaccine. mRNA vaccines, for instance, can be produced more quickly than traditional inactivated or live-attenuated vaccines.
Distribution Challenges and Strategies
After production, vaccines must be efficiently distributed—a task that involves coordination between governments, health organizations, manufacturers, and logistics companies. Distribution requires cold chain systems, which are temperature-controlled supply chains necessary for vaccine storage and transport. Some vaccines, such as the Pfizer-BioNTech COVID-19 vaccine, require ultra-cold storage at temperatures around -70°C, posing additional logistical challenges, especially in low-resource settings.
National and global health agencies create distribution strategies based on priority groups. High-risk populations, such as healthcare workers, the elderly, and people with underlying health conditions, are often vaccinated first. This phased approach helps to maximize the public health benefit, especially during limited supply periods.
Technology plays a significant role in distribution logistics. Real-time tracking systems monitor vaccine shipments, ensuring they remain within required temperature ranges. Barcode scanning and electronic health records help manage inventory and track immunization coverage.
Global Equity and Access
One of the biggest challenges in vaccine distribution is ensuring equitable access across countries and communities. Wealthier nations often have greater resources to secure vaccine supplies, while low-income countries may struggle with procurement and distribution infrastructure. Initiatives like COVAX, led by Gavi, the Vaccine Alliance, WHO, and other partners, aim to provide equitable vaccine access worldwide.
In addition to supply issues, vaccine hesitancy can hinder distribution efforts. Public health campaigns are crucial to educate communities, build trust, and encourage vaccine uptake.
Conclusion
Vaccine production and distribution is a highly coordinated global effort that blends science, technology, and logistics. From the lab bench to the arm of a patient, every step is vital in ensuring that vaccines are safe, effective, and available to those who need them most. As new diseases emerge and global health threats continue to evolve, the ability to rapidly produce and equitably distribute vaccines remains one of our strongest tools in protecting public health.
Our Products
-
Metformin 500mg
$0.80 / Per Pill
-
Testosterone Gel
$4.00 / Per Sachet
-
Lisinopril 20mg
$1.00 / Per Pill